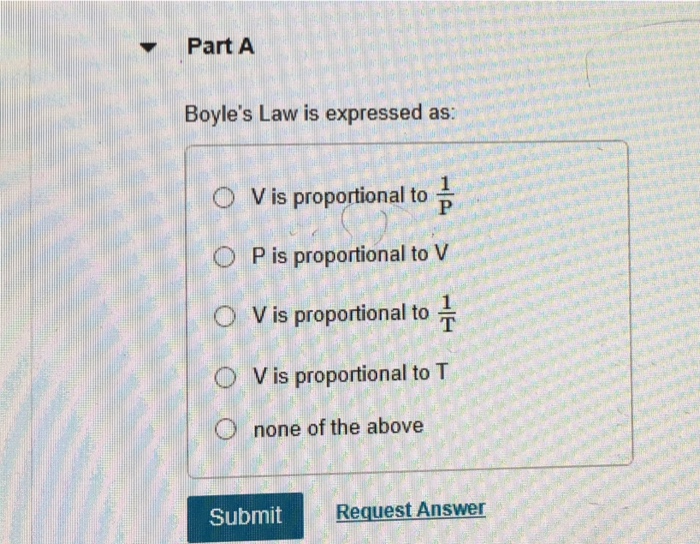
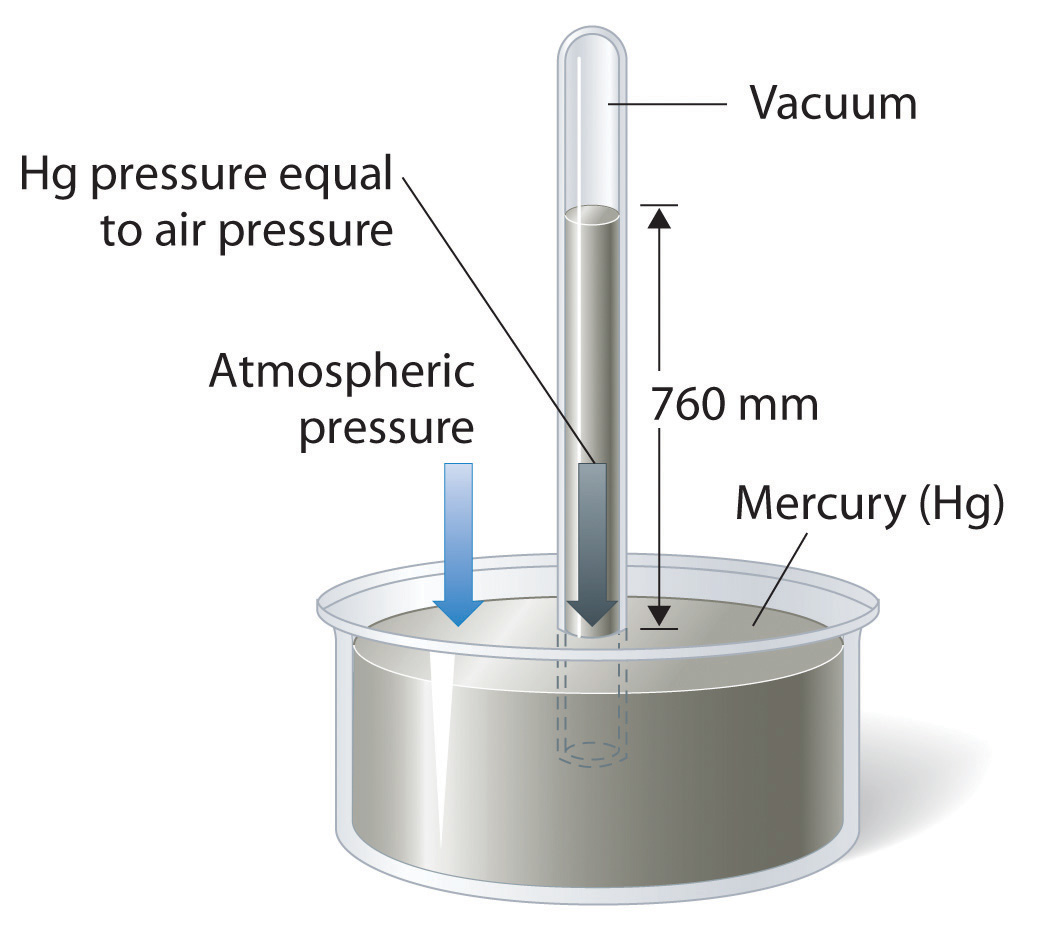
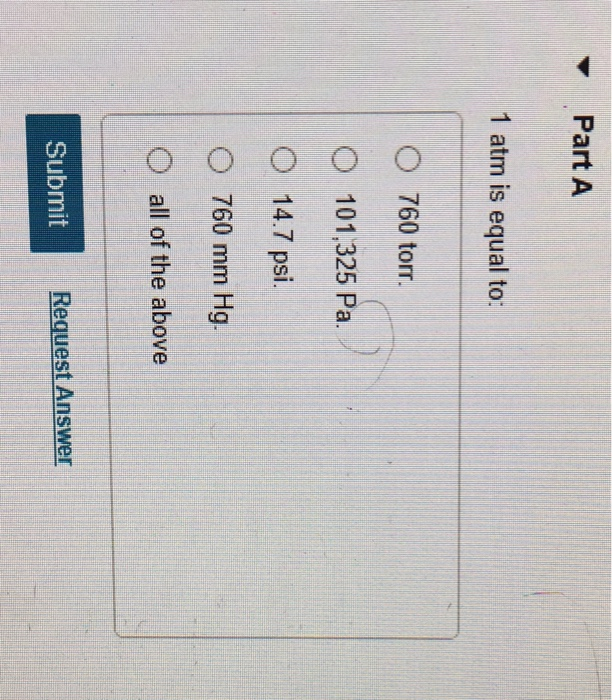
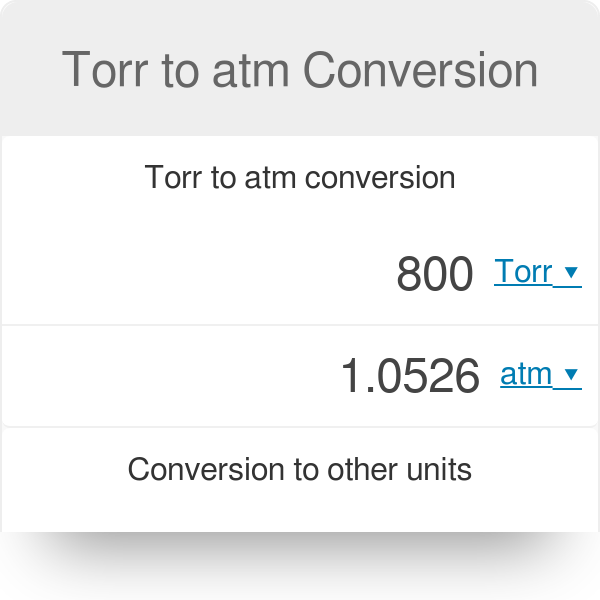
The atmospheric pressure on a mountain is 550 mmHg and 1 atm is equal to 760 mmHg. What is the - Brainly.com

SOLVED: 21. Calculate the value of R, the gas constant; when 3 moles of He gas is placed under a pressure of 1 atm at OC with a volume equal to 67.2

Doubt: Question number 6 how to solve this with units Chapter: Thermodynamics - Subject: Chemistry - Course: NEET Course - Complete Syllabus

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

GASES. Identify the abundances of the naturally occurring gases in the atmosphere. Describe the historical development of the measurement of pressure. - ppt download