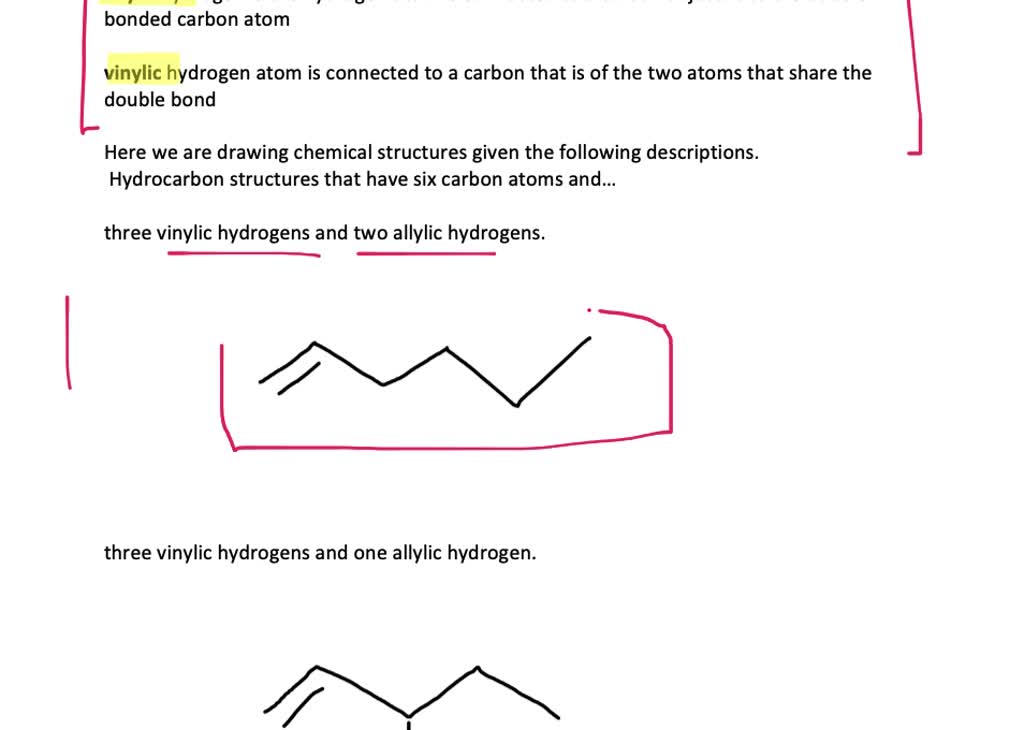
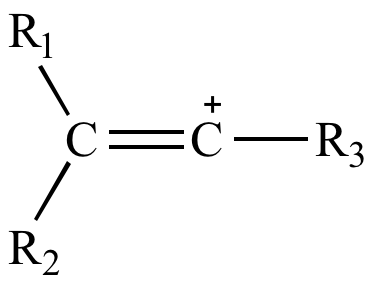
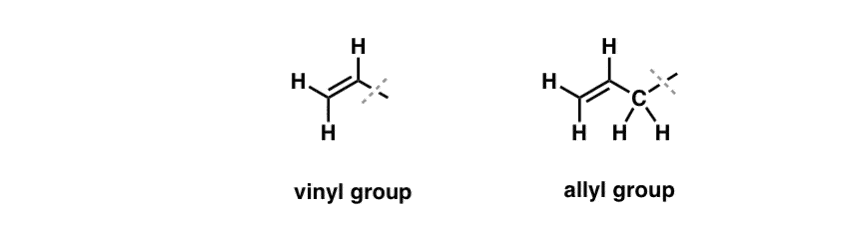
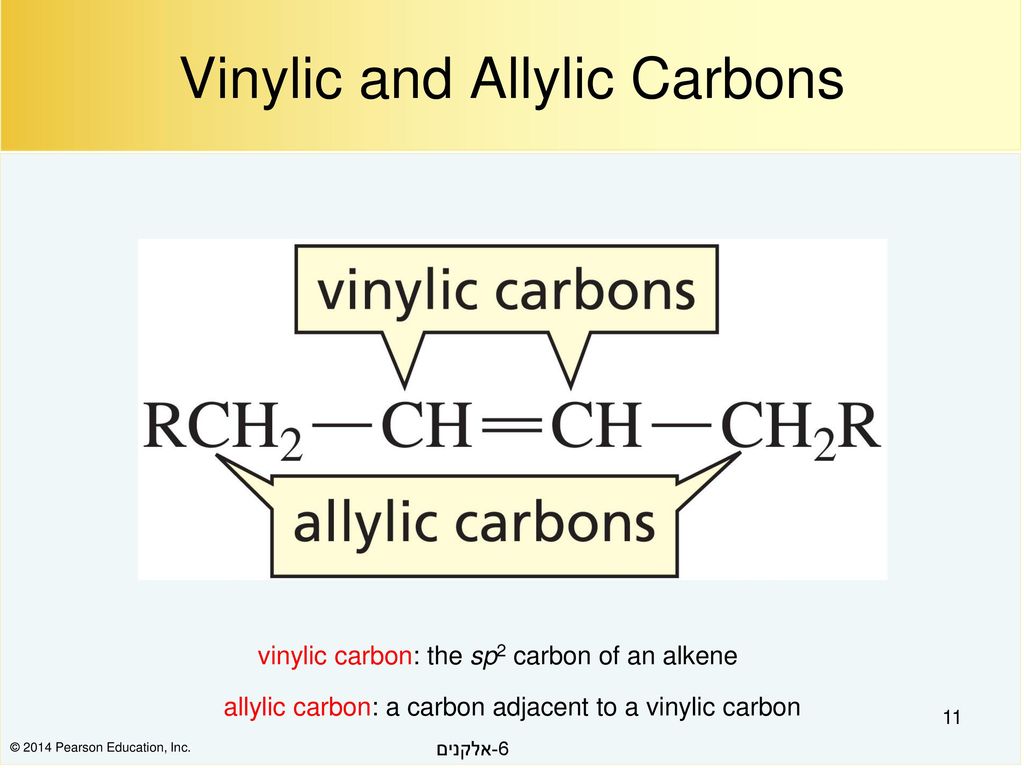
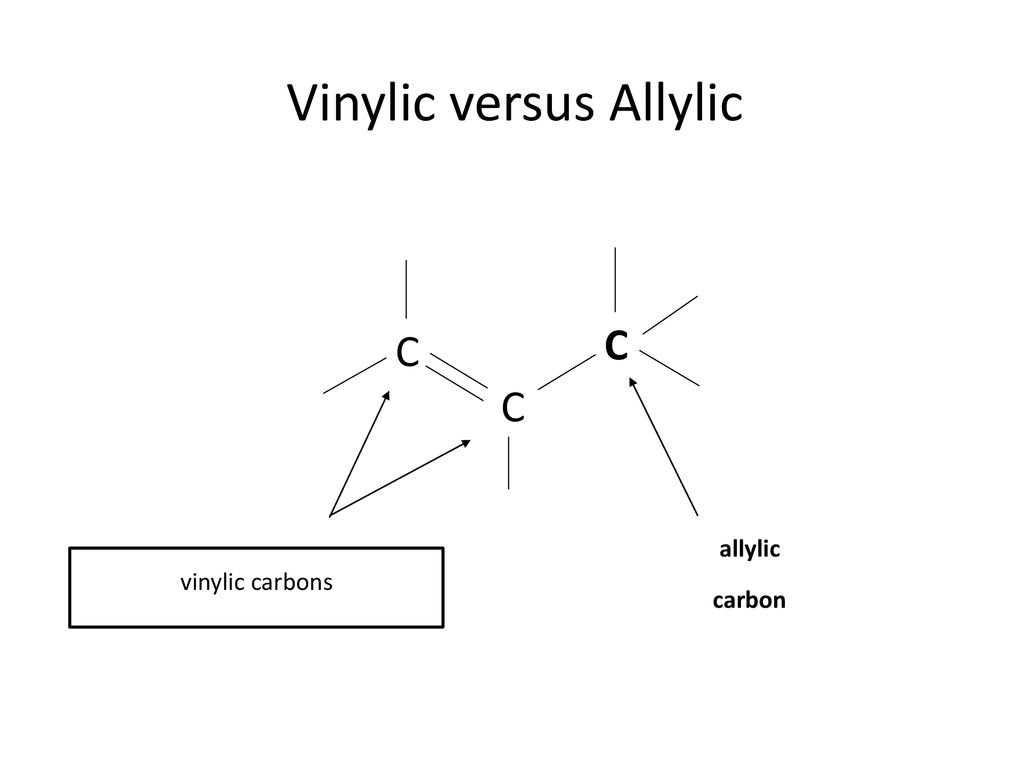
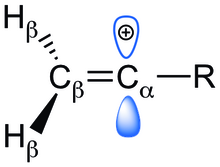
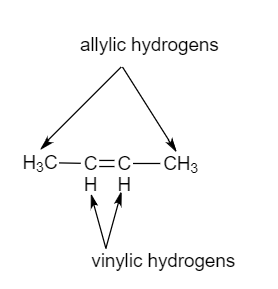
what do you mean by allylic carbon and vinylic carbon with diagram - Chemistry - Haloalkanes and Haloarenes - 3567919 | Meritnation.com

organic chemistry - GOC allylic,vinylic, benzylic positions, carbocation stability - Chemistry Stack Exchange
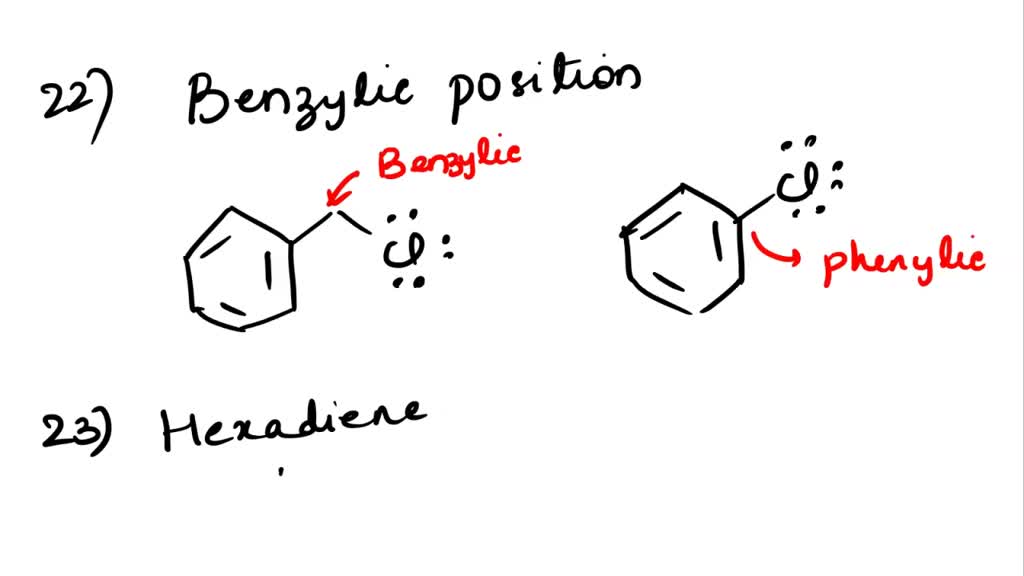
SOLVED: '22. Any carbon atom attached directly to benzene ring is called Vinylic position B) Allylic position C) Phenylic position D) Benzylic position E) Acidic position 23. In Birch reduction, the aromatic
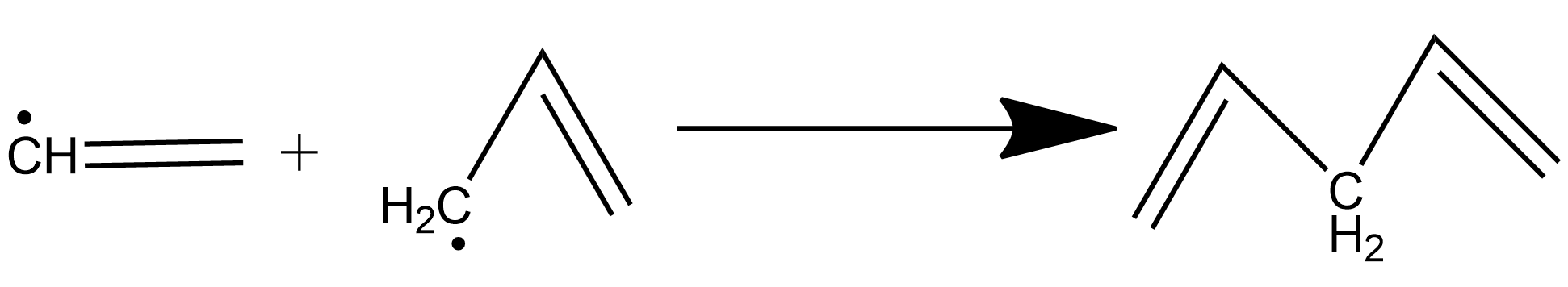
When vinyl allyl are joined to each other, we get:A. conjugated alkadieneB. cumulative alkadieneC. isolated alkadieneD. allenes

How Vinyl, Allyl and Homoallyl Describe the Position of an Alkene Relative to Another Functional Group - Chemistry School

Rare Example of Nucleophilic Substitution at Vinylic Carbon with Inversion: Mechanism of Methyleneaziridine Formation by Sodium Amide Induced Ring Closure Revisited | Journal of the American Chemical Society
Can we classify the vinylic carbon in alkene to be primary, secondary or tertiary? If yes, how? - Quora
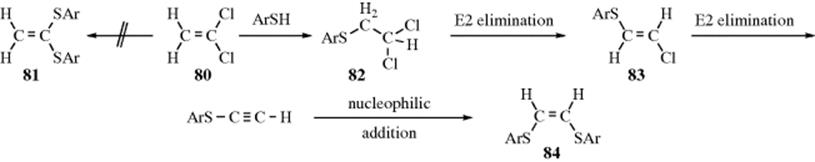
Nucleophilic Substitution at an Aliphatic Trigonal Carbon: The Tetrahedral Mechanism - Aliphatic Substitution, Nucleophilic and Organometallic - Introduction - March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 7th Edition (2013)
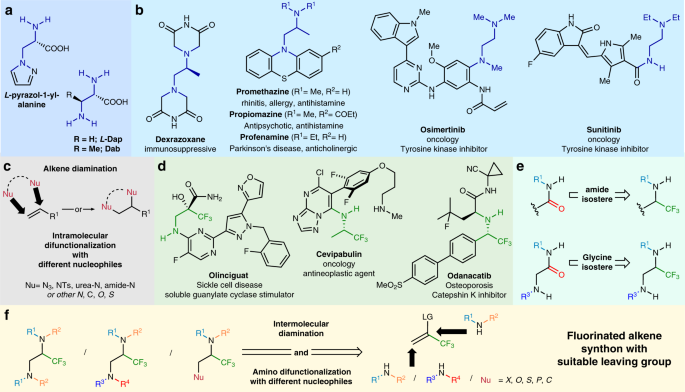
Vicinal difunctionalization of carbon–carbon double bond for the platform synthesis of trifluoroalkyl amines | Nature Communications